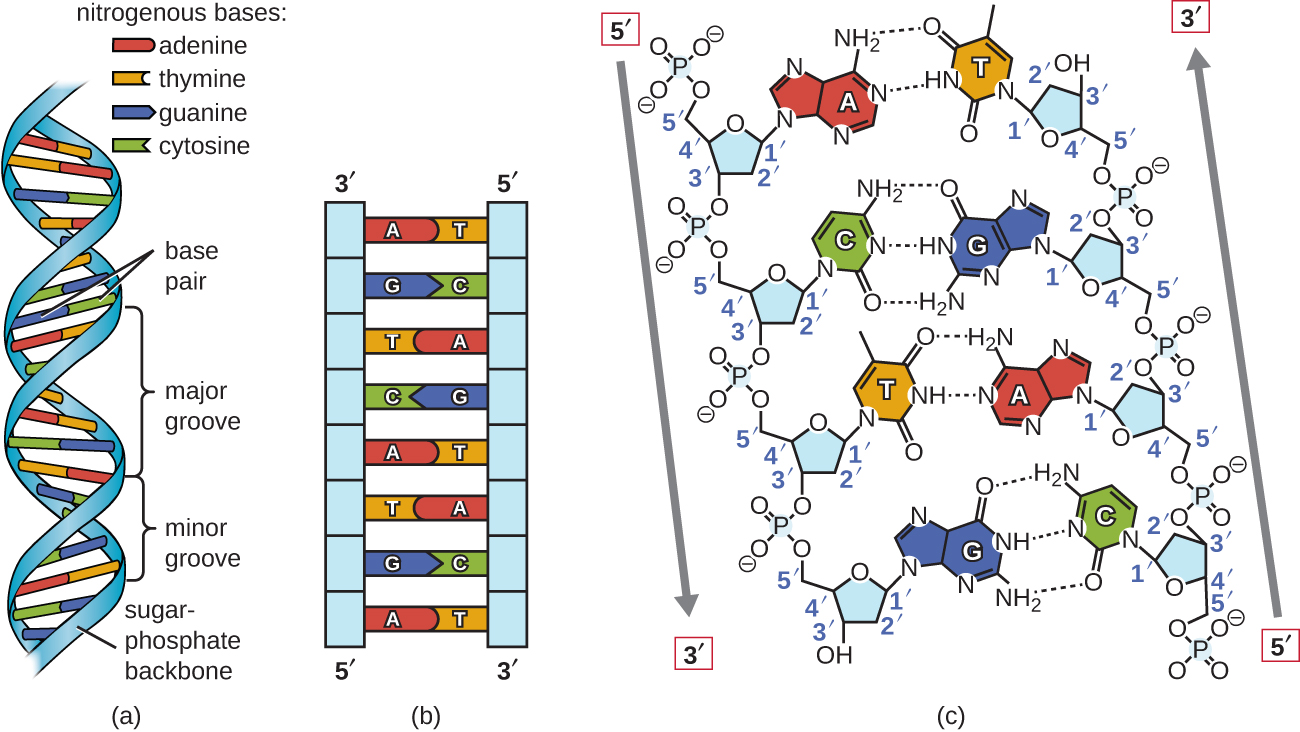
Of course, the point of the genetic information in DNA is to encode the production of proteins that can then carry out the functions that define cellular life. Because of base pairing, each strand of DNA contains all the information necessary to make a complete exact copy of its complementary strand. Conversely, the same strand of DNA is read over and over to make the RNA, and since there are only two copies of each chromosome (a chromosome is a single double-stranded DNA molecule) in a cell, the ability to maintain the integrity of the DNA is crucial. Its long-term stability is not particularly important because when it is made, usually many copies are made at the time, and it is only needed while the cell needs to make the protein it encodes. Therefore, RNA needs to be easily read, meaning that the bases need to be accessible, and not locked to a complementary strand. This difference is important to their function: RNA is a temporary information transfer molecule for a particular gene, DNA is the permanent repository of all genetic information needed to make an organism. However, in the cell, most RNA is single-stranded, and most DNA is double-stranded. 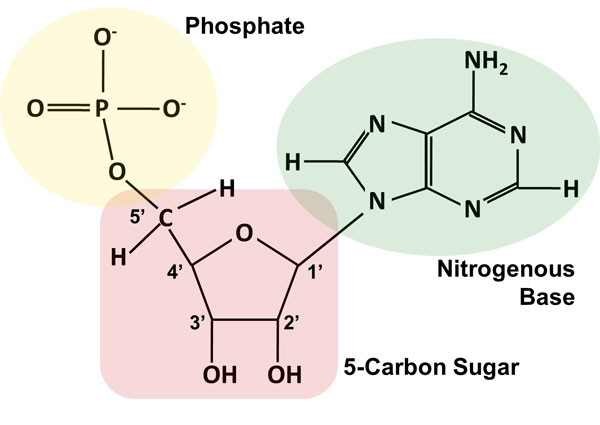
Nucleic acids, the long polymers of nucleotides, exist in either single or double stranded forms in vitro. This specific base-pairing, known as Chargaff’s rules, is the basis for life: base-pairing is needed to make DNA double stranded, which gives an organism a built-in backup of genetic information and it is also the basis for transforming that information into proteins that form the bulk of a cell. Note that while one extra hydrogen bond does not appear to be particularly significant, the attraction between G-C is 50% stronger than between A-T, and over long stretches of DNA, areas high in G-C content are significantly more difficult to unzip (separate strands) than areas high in A-T pairs. Not only do they interact, but they interact with great specificity and consistency: adenines base-pair with thymines (or uracils) through two hydrogen bonds, while guanines interact with cytosine through three H-bonds. The bases will then likely interact with the bases of other nucleotides, whether part of another nucleic acid strand or free-floating. Both are constructed from nucleotides like adenosine triphosphate (C). (A) DNA and (B) RNA differ by the presence of -OH on the 2-carbon of ribose but not deoxyribose and the use of uracil in RNA instead of thymine. In contrast, DNA samples can be safely stored in aqueous buffer in a refrigerator, or in a freezer for longer-term storage.\). The problem of RNA decomposition is compounded by the fact that RNAase enzymes, which catalyze RNA hydrolysis, are present on the surface of human skin and are very stable, long-lived, and difficult to destroy. Researchers working with RNA have to be careful to store their samples at very cold temperatures, preferably freeze-dried or precipitated in ethanol, to avoid hydrolysis. The 2' hydroxyl group is right next to the electrophilic phosphorus atom, poised in a good position to make a nucleophilic attack, breaking the RNA chain and forming a cyclic phosphate diester intermediate (see figure below). RNA nucleotides, unlike the deoxynucleotides of DNA, have a hydroxyl group at the neighboring 2' carbon. Why does hydrolysis occur so much more rapidly in RNA than in DNA? The answer has everything to do with the lowered entropic barrier to the reaction (you might want to quickly review the concept of entropy at this point). This does not present a physiological dilemma, because the function of RNA is to encode genetic information on a temporary rather than long-term basis. Unlike DNA, RNA is quite vulnerable to spontaneous hydrolysis in aqueous solution. Enzymes that hydrolyze the phosphate diester bonds in DNA are called nucleases, and we will learn more about them in section 9.8.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
